
Press Releases
- Shilpa Medicare Backs US Biotech Alveolus Bio to Accelerate Next-Gen Lung Disease Therapies
- Shilpa Biocare, a wholly owned subsidiary of Shilpa Medicare enters into a strategic partnership with Orion Corporation (“Orion”), Finland
- Launch of 2nd NDA Bortezomib for injection3.5mg 1.4ml in US
- US Approval for Varenicline Tablets, 0.5 mg and 1 mg
- Europe (AGES, Austria) Inspection of Shilpa Biologicals Pvt Ltd (SBPL), a wholly owned subsidiary of SML
- Approval by European Medicines Agency, for Shilpa Medicare Ltd
- Shilpa Pharma Lifesciences Limited, Unit-1, Received EIR from USFDA
- EIR from USFDA, for Shilpa Medicare Ltd. Unit VI, Bengaluru
- Initial Authorization for Rivaroxaban Orodispersible Films from Europe
- Initial Authorization for Rivaroxaban Orodispersible Films from EuropStake Sale in Sravathi Advance Process Technologies
- Shilpa Biologicals Pvt Ltd signs strategic development partnership with mAbTree Biologics AG for a Novel Immunooncology Asset
- Clinical trial Announcement for SML 007
- Eurasia (EAEU) GMP Approval of Shilpa Medicare Ltd. Unit IV, Jadcherla, Telangana and Unit VII
- Europe Approval for Tadalafil Orodisperible Films
- Europe GMP Approval of Shilpa Medicare Ltd. Unit 4, Jadcherla, Telangana
- Important milestone for the CDMO business of SML
- Indian Regulatory Authority - SEC approval for conducting Phase III Clinical trials for Recombinant Human Albumin (rHA) 20%
- Shilpa Biologicals Pvt Ltd signs strategic development partnership with mAbTree Biologics AG for a novel immuno- Onco asset
- Shilpa Biologics Pvt Ltd (“SBPL”) receives first international market approval for Adalimumab 40 mg in Morocco
- Shilpa Medicare announces acceptance by USFDA of NDA filed by its CDMO partner Unicycive - OLC
- Shilpa Medicare announces successful outcome of Phase 3 studies of SMLNUD07 – NorUDCA
- Shilpa Pharma Lifesciences Limited, Received CEP from EDQM for API, Desmopressin
- Shilpa Pharma Lifesciences Limited, Received CEP from EDQM for API, Nifedipine
- Shilpa Pharma Lifesciences Limited, Received CEP from EDQM for API, Octreotide
- Shilpa Pharma Lifesciences Limited, Received CEP from EDQM for API, Teriflunomide
- Shilpa Pharma Lifesciences Limited, Received CEP from EDQM for API, Ursodeoxycholic Acid
- Shilpa Pharma Lifesciences Limited, Unit 2, Raichur, Karnataka has cleared the ANVISA-Brazil GMP inspection
- SML’s CDMO customer receives Fast-Track Designation for its investigational drug candidate
- SML’s CDMO customer receives Fast-Track Designation for its investigational drug candidate
- Unit 2, Raichur, Karnataka has cleared the COFEPRIS-Mexico GMP
- USFDA Approval of NDA IMKELDI (Imatinib Oral Solution) Filled by our partner Shorla Oncology
- Acquisition of Pilnova Pharma Inc - A Wholly owned Subsidiary
- Approval by TGA, Australia for Shilpa Medicare Ltd. Unit VI, Bengaluru
- Europe Approval for Amifampridine Tablets, 10 mg
- Europe Approval for Varenicline Tablets, 0.5mg and 1mg
- Grant of Code “J” for Pemetrexed Injection, 100mg 10 ml, 500 mg 50 ml, 1000 mg 100 ml
- Marketing Authorization for Adalimumab 40 mg 0.4 ml Injection
- Shilpa Medicare Limited Launches Lenvatinib Capsules (12mg, 18mg & 24 mg) in India under the brand name Lenshil
- Shilpa Medicare Limited Launches Pemetrexed Injection (10 ml, 50 ml, 85 ml, and 100 ml sizes) in India
- Shilpa Medicare Limited receives US FDA approval for psoriatic arthritis drug
- Shilpa Medicare Limited, Analytical Services Division (Unit 7, Nacharam, Hyderabad), US FDA GMP
- Shilpa Medicare Ltd, Unit IV, Jadcherla, Telangana has cleared ANVISA Brazil, GMP inspection
- Shilpa Pharma Lifesciences Ltd, Unit II, Raichur, Karnataka has cleared PMDA Japan, GMP inspection
- Stake sale of 4% of its Stake held in Sravathi Advance Process Technologies Private Limited
- UK‐MHRA Approval for Betahistine Dihydrochloride Orodispersible Films, 24 mg
19 Sept 2025 :
Dear Our Trade Distributors, Stockiest & Business Associates,
We wish to inform you that, in accordance with the latest directives from the GST Council and the National Pharmaceutical Pricing Authority (NPPA), significant changes to the Goods and Services Tax (GST) and Drugs (Prices Control) Order (DPCO) will come into effect starting 22nd September 2025. In line with these guidelines, Shilpa Medicare Ltd is publishing the Product Information like Brand Name, Generic Name, PTS, PTR, Old MRP, Old GST Rate & Revised GST Rate along with Revised MRP effective from 22nd Sep 2025 to ease the Business Operation of Our All Associates along with Awareness on Pricing & Tax Structure.
It is mandatory to pass the Benefit of Reduction of GST rate to the ultimate customer. Attached is the Guideline & Product Details:
19 Sept 2025 :
Dear Our Trade Distributors, Stockiest & Business Associates,
We wish to inform you that, in accordance with the latest directives from the GST Council and the National Pharmaceutical Pricing Authority (NPPA), significant changes to the Goods and Services Tax (GST) and Drugs (Prices Control) Order (DPCO) will come into effect starting 22nd September 2025. In line with these guidelines, Shilpa Medicare Ltd is publishing the Product Information like Brand Name, Generic Name, PTS, PTR, Old MRP, Old GST Rate & Revised GST Rate along with Revised MRP effective from 22nd Sep 2025 to ease the Business Operation of Our All Associates along with Awareness on Pricing & Tax Structure.
It is mandatory to pass the Benefit of Reduction of GST rate to the ultimate customer. Attached is the Guideline & Product Details:
19 Oct 2023 :
Shilpa Medicare Limited has launched ORAAL® for the first time in India, a revolutionary product designed and naturally derived to provide relief and treatment induced by Radiotherapy or Chemotherapy. This product has received approval from the Office of the Directorate of Ayurveda and Unani Services (a division of the Ministry of Aayush) and is globally patented.
ORAAL® is endorsed by the Indian Dental Association (IDA) and is suitable for all Mucositis grades. It offers a flexible and convenient oral spray solution, reducing the need for chemical mouth rinses and ensuring precise dosing without side effects. With key ingredients like Curcuma Longa, Honey, Glycerine, and Purified Water, ORAAL® provides soothing relief in a convenient spray form. This throat spray, presented in a 20ml bottle, targets pro-inflammatory proteins, contaminants, and impurities, offering a threefold action for optimal results.

05 July 2023 : We are delighted to announce that Shilpa Biologicals Private Limited (SBPL), a wholly owned subsidiary of Shilpa Medicare Limited (SML), has received the Marketing Authorization Permission for its Adalimumab 40 mg/0.4mL injection in prefilled syringe (PFS), a biosimilar of Adalimumab higher concentration formulation (100mg/mL) in India. This is used for the treatment of Rheumatoid Arthritis (moderate to severe active RA & severe active and progressive RA) for which the phase 3 clinical trial has been successfully completed in Q3 2022. This formulation will contribute to increased patient comfort based on reduced injection volume. Shilpa Biologicals' extensive research and development efforts have resulted in the successful development of a high concentration biosimilar that matches the safety, purity, and potency profiles of the reference product. This achievement underscores the company's commitment to development of biosimilars and its mission to improve global healthcare outcomes. SBPL has been working on development of biosimilars catering to different therapeutic areas such as Immunology, Oncology, Ophthalmology, among which, Adalimumab is the first Biosimilar from Shilpa Biologicals Private Limited
23 May 2023 : Shilpa Medicare’s New Drug Application for Pemetrexed Injection 1000 mg/100 ml, 500 mg/50 ml & 100 mg/10 ml, Ready to Use formulation is approved by US FDA.This is the first NDA product approval received by the Company under 505(b)(2) application, which is a unique formulation supplied as ready to use product solution. This product is required to be administered intravenously without any further dilution. The product is stable at room temperature.The company has developed Pemetrexed formulation as liquid/ ready to use injection, against Lyophilized powder formulation of reference, ALIMTA®. The product approval has been received for the application filed from a third party manufacturing site.Pemetrexed injection is used in the treatment of Non-small cell lung cancer & Malignant pleural mesothelioma as approved in the Prescribing Information
11 Sep 2022 :
Approval of Capecitabine Dispersible Tablets 150 mg, 500 mg & 1000 mg
Shilpa Medicare has received approval of Capecitabine Dispersible Tablets 150 mg, 500 mg & 1000 mg for commercial distribution for the first time in India. The product will be commercialized in India under the brand name CAPEBEL DT. The company has developed additional strength (1000 mg) of Capecitabine oral formulation which can be taken as dispersion in water. Introduction of additional strength in the form of dispersible tablets will ease the administration of patients, as they usually suffer from dysphagia. Capecitabine Dispersible Tablets are approved for the treatment of patients with metastatic colorectal cancer and for the treatment of metastatic breast cancer after failure of paclitaxel and anthracycline containing chemotherapy regimen.

27 Sep 2022 : Shilpa’s Tranexamic Acid Spray (Hemostatic Spray) is approved by CDSCO (DCGi). Shilpa would like to announce its successful innovation of first in world topical haemostatic spray of Tranexamic Acid has received CDSCO approval. This novel formulation of spray is simple, easy to carry, convenient to use and a lifesaving tool. Clinically proven in humans and validated in robust swine trauma model. The product is approved for the usage of reducing or preventing haemorrhage and to reduce the need of replacement therapy during accidental and domestic injury. • Quick hemostasis- controls moderate to severe bleeding within minutes • Advanced Hemostat with dual mechanism of action • Cuts, lacerations, puncture, stab wounds & combat wounds • Emergency Bleeding & pre-hospital trauma life support • As potential life-saving tool in first-aid box for traffic/road accidents and battlefield injuries. • Anti-bacterial properties of chitosan reduce micro-organisms during transport • beneficial in civilian prehospital care, emergency medical systems (EMS) and ambulance supply. • Less chances of re-bleeding during transport to hospital
12 Aug 2022 :
CPhI Worldwide 2022 (Nov. 1st – 3rd, Frankfurt).
We are pleased to announce the presence of Shilpa Medicare at CPhI Worldwide 2022, the must attend pharma industry event of the year. If you are seeking new business opportunities and a credible delivery and innovation partner, please visit us at Stand #12D62. We look forward to seeing you!
For more information on our CDMO operations and to schedule a meeting, please contact us at [email protected]

12 July 2022 : Shilpa Medicare Ltd, Unit VI, situated at Dabaspet, Bengaluru, Karnataka State, has been issued GMP Certificate by UK MHRA. The facility was inspected by MHRA from 18 Apr 2022 to 20 Apr 2022. . The facility is involved in manufacturing and testing specialized dosage forms like Orodispersible Films and Transdermal systems. It is backed up with strong Research and Development of orodispersible films and transdermal systems. The GMP facility is equipped with state of art infrastructure to meet commercial requirements from a global perspective
26 May 2022 :
The Prestigious PEL -HEMSI AWARD presented to Polymer Research & Technology Group of Shilpa Medicare Limited on 26th May 2022 at 13th International High Energy Material Conference and Exhibits at Chandigarh India. . Dr. Ganugapati Satyanarayana - Senior vice President and Head at SML received the award from distinguished directors of DRDO and ISRO Establishments.at 13th International High Energy Material Conference & Exhibits on 26th May 2022.
The Research & Technology Team established a One -Pot scalable process to produce the anionic HTPB as per desired specifications for the Defence application. The produced anionic HTPB has a narrow PDI, low vinyl content and stable hydroxyl end-group compared with radical HTPB.
SML optimized the process at a pilot scale using designed of experimental studies, further designed & commissioned dedicated manufacturing facility to operate at 25 -50 kg batch sizes. The current optimized process and facility guide the futuristic production of anionic HTPB tonnage levels.
This paved way for SML to create niche space in creating skill sets & facilities for scale up a highly skilled anionic polymerization technique in India first time

26 Jan 2022 :
Shilpa Medicare Limited, Unit VII (Analytical Services) situated at Nacharam, Hyderabad location has been issued "NABL Accreditation" . This site is involved in physico-chemical testing of raw materials (excipients), packing materials, drug substance, drug products. The facility is equipped with state of art analytical instruments to perform testing. The site already obtained testing permission from State Drug Control Administration in Form 37.”.

17 Sep 2021 :
Shilpa Medicare Ltd has received “ Best District Export Excellence Award” from FKCCI on 15th September 2021.

31 Aug 2021 :
“Company has received DCG(i) approval for 2-Deoxy-D-Glucose (Bulk & Oral Powder). The bulk is manufactured by Shilpa Medicare Limited, API facility in Raichur, Karnataka. The Oral Powder is manufactured by its subsidiary, Shilpa Therapeutics, facility in Hyderabad, Telangana. 2-Deoxy-D-Glucose Oral Powder is approved for the treatment of adjunct therapy in moderate to severe COVID-19 patients. The product will be available with a brand name ‘2-DGSHIL’.”
03 Aug 2021 :
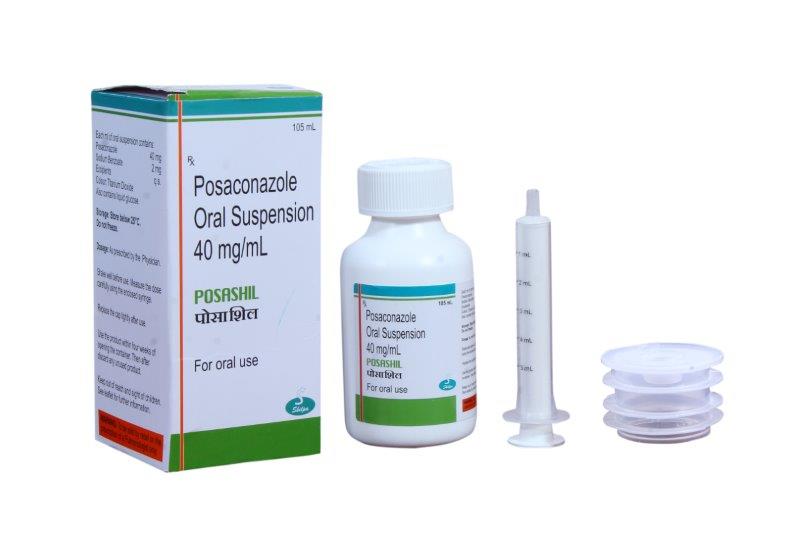
“Shilpa Medicare Ltd. has launched Posaconazole Suspension with unique applicator, first of its kind highly convenient pack under the brand name “Posashil” ”
Posashil Suspension (40mg Posaconazole / mL in 105 mL pack) are indicated for the treatment of patients with Mucormycosis and Prophylaxis of Invasive Fungal Infections, Oropharyngeal Candidiasis etc.
Posashil has been launched with a unique packing, a unique Oral Dispenser, Adaptor unlike the traditional Posaconazole suspension brands is being offered with a spoon or a measuring cup. This makes Posashil a unique product – which ensures no spillage of medicine and convenient to consume by the patient.
25 June 2021 :
“Shilpa Medicare has received an in-principle approval from Defence Research & Development Organisation (DRDO) to manufacture and sale of 2-Deoxy-D-Glucose (2DG).
2DG has been given emergency approval by the Drug Controller General of India (DCGI) for COVID-19 patients in the country. Shilpa Medicare is only the second company in India to have entered into similar arrangement with DRDO”.
15 Jun 2021 :
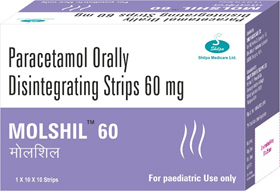
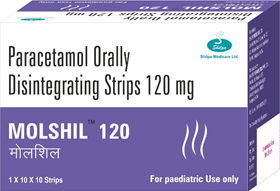
“Shilpa Medicare Ltd., introduces World’s First Paracetamol in Orally Disintegrating Strip ”
Kids melt our heart with their naughty actions and when they have fever entire family worries about them. Melt their fever like they melt our hearts with Molshil.
Molshil is available in 60 mg & 120 mg strengths with tangy orange flavour.
Molshil addresses all these needs and improves infant, parent quality of life.
May 2021 :
“Shilpa Medicare Ltd, has received the RU GMP certificate from the Russian MOH”


May 2021 :
“Shilpa Biologicals Pvt Ltd, (SBPL) the wholly owned subsidiary of Shilpa Medicare Ltd. has entered in a 3-year definitive agreement with Dr. Reddy's Laboratories for the production-supply of Sputnik V vaccine”
April 26 2021 : Shilpa Pharma, Inc. Sues Novartis Pharmaceutical Corporation for Patent Infringement for “Fingolimod Polymorph and Their Processes”.
March 3 2021 :
“Shilpa Medicare Ltd., has launched India’s first women intimate cleansing spray under the brand name “Swatchshil”.
“SwatchShil” intimate Cleansing Spray is a Soap-free and alcohol-free solution with a pH of 3.7± 0.5, ideal for the Women intimate area, helps in maintaining the optimum pH level and avoids vaginal discomfort and effectively maintain intimate hygiene.
”


Feb 15 2021 :
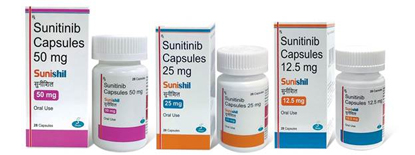
“Shilpa Medicare Ltd has launched Sunitinib Capsules 12.5 mg, 25 mg & 50 mg in Indian market under brand name “SUNISHIL”. Sunitinib Capsules are indicated for the treatment of Gastrointestinal stromal tumour (GIST), Metastatic renal cell carcinoma (MRCC) & Pancreatic neuroendocrine tumours (pNET). SUNISHIL is a therapeutic equivalent of the brand leader SUTENT. ”
Feb 12 2021 :
“Shilpa Medicare Limited has launched Dimethyl Fumarte Delayed Release Capsules 120 mg & 240 mg in India under the brand name “DMFShil”. Shilpa’s DMFShil is a therapeutic equivalent of TECFIDERA (brand leader). DMFShil is used in the treatment of Relapsing remitting multiple sclerosis. ”

Aug 26 2020 : “Shilpa Medicare Limited has successfully launched Azacitidine 25 mg/ml powder for suspension for injection in Europe through its partners. Azacitidine 25 mg/ml powder for suspension for injection is a generic equivalent of VIDAZA (brand leader) approved for the treatment of adult patients who are not eligible for haematopoietic stem cell transplantation (HSCT) with myelodysplastic syndromes, chronic myelomonocytic leukaemia & acute myeloid leukaemia. ”
Aug 26 2020 :
“Shilpa has successfully launched for the first time in the world, ready to drink “Green Tea Film”. Shilpa's Green Tea film contains 120 mg of green tea extracts with 98% Polyphenol content. Green Tea is also known for its antioxidant properties. Green Tea is useful for the weight reduction and beneficial effects on LDL cholesterol regulation. ”
For more details please do visit on www.shilpaotc.com .

June 30 2020 :
“Shilpa Medicare Limited launches the first Indian branded generic of Axitinib Tablets 1 mg & 5 mg, an anti-cancer drug with a brand name AXISHIL.
AXISHIL is used to treat patients with ‘First-Line Advanced Renal Cell Carcinoma’ and ‘Second-Line Advanced Renal Cell Carcinoma’.
Shilpa’s AXISHIL is the generic equivalent & bioequivalent of the brand leader INLYTA.”
June 2 2020 :
“Shilpa Medicare Limited launches the Indian branded generic of Ibrutinib Capsules 140 mg, an anti-cancer drug with a brand name IBRUSHIL.
IBRUSHIL is used to treat patients suffering from Chronic Lymphocytic Leukemia (CLL), Mantle Cell Lymphomas (MCL) and other related cancers.
Shilpa’s IBRUSHIL is the generic equivalent & bioequivalent of the brand leader IMBRUVICA.”
May 23 2020 :
“Shilpa Medicare Limited has received CDSCO -New Delhi Permission for manufacturing and marketing of Lenvatinib capsules 4mg and 10mg in India. Shilpa Medicare Limited is the “First Company” to obtain Generic approval for Lenvatinib Capsules 4mg and 10mg in India.
Lenvatinib capsules 4mg and 10mg is a generic equivalent & bioequivalent of reference product, Lenvima, used in the treatment of patients with locally recurrent or metastatic, progressive, radioactive iodine-refractory differentiated thyroid cancer and for the first line treatment of patients with unresectable hepatocellular carcinoma.
”
April 12 2020 :
“Shilpa Medicare Limited has launched first branded generic anti-cancer drug, Dasatinib with all dosage strengths 20 mg, 50 mg, 70 mg & 100 mg under brand name – DASASHIL.
The drug substance patent on Dasatinib was owned by Bristol-Myers-Squibb and was expired on 12 April 2020.
Dasatinib is used for the treatment of Chronic Myeloid Leukaemia.
DASASHIL will revolutionise the treatment by ensuring to make therapy available for more Indian patients due to increased affordability. DASASHIL is being manufactured and supplied from the state-of-art US FDA approved facility.
”




Jan 13 2020 : “Shilpa’s Formulation (Oral Solids & Injections) manufacturing facility located at Jadcherla, Telangana, has been issued EU GMP certificate by AGES, Austria for the inspection done during 13 to 17 Jan 2020.”
Fab 03 2020 : “Shilpa Medicare Limited has successfully completed USFDA inspection at both API sites located at Raichur (Unit-1 & Unit-2) between 3rd and 7th Feb, 2020 with ZERO 483s.”
Jan 30 2020 :
“Shilpa Medicare Limited has received U.S Food and Drug Administration tentative approval for its ANDA, Pirfenidone Tablets, 267 mg and 801 mg on 30 Jan 2020. The ANDA is filed as ‘First to File’ submission on NCE -1 date.
Pirfenidone Tablets, 267 mg and 801 mg is a generic equivalent of reference listed drug (RLD) Esbriet Tablets, 267 mg and 801 mg, of Hoffmann La Roche Inc. used in the treatment of idiopathic pulmonary fibrosis as recommended in the label approved by FDA.”
Aug 08 2019 : “Shilpa Medicare Limited has received CEP from EDQM for Tranexamic Acid (R0-CEP 2018-048-Rev 00.)”
May 16, 2019 :
“Shilpa Medicare Limited formulation division has received U.S Food and Drug Administration final approval for its ANDA, Docetaxel Injection USP, 20 mg/mL, 80 mg/4 mL (20 mg/mL), and 160 mg/8 mL (20 mg/mL) on 16.05.2019.
Docetaxel Injection USP, 20 mg/mL, 80 mg/4 mL and 160 mg/8 mL is a generic equivalent of reference listed drug (RLD) TAXOTERE used in the treatment of Breast Cancer, Non-Small Cell Lung Cancer, Prostate Cancer, Gastric Adenocarcinoma, Head and Neck Cancer as recommended in the label approved by FDA.”
May 15, 2019 :
Shilpa Medicare Ltd formulation division has received U.S Food and Drug Administration final approval for its ANDA, Zoledronic Acid Injection, 4 mg/5 mL on 15.05.2019.
Zoledronic Acid Injection, 4 mg/5 mL is a generic equivalent of reference listed drug (RLD) ZOMETA. It is a bisphosphonate indicated for the treatment of Hypercalcemia of malignancy, Patients with multiple myeloma and patients with documented bone metastases from solid tumors, in conjunction with standard antineoplastic therapy as recommended in the label approved by FDA.
April 18, 2019 :
Shilpa Medicare Ltd has received U.S. Food and Drug Administration approval for its ANDA, Busulfan Injection, 60 mg/10 mL.
Busulfan Injection, 60 mg/10 mL is a generic equivalent of reference listed drug (RLD), Busulfex Injection, 60 mg/10 mL, used in the treatment of patients with chronic myelogenous leukemia as recommended in the label approved by FDA.
Feb 22, 2019 :
Shilpa Medicare Ltd has received U.S. Food and Drug Administration approval for its ANDA, Gemcitabine for Injection USP, 200 mg/vial and 1 g/vial.
Gemcitabine for Injection USP is a generic equivalent of reference listed drug (RLD), GEMZAR used in the treatment of ovarian cancer, breast cancer, non-small cell lung cancer & pancreatic cancer as recommended in the label approved by FDA.
Jan 17, 2019 :
Shilpa Medicare Ltd has received U.S. Food and Drug Administration approval for its ANDA, Imatinib Mesylate Tablets, 100 mg and 400 mg.
Imatinib Mesylate Tablets is a generic equivalent of reference listed drug (RLD), Gleevec Tablets, 100 mg and 400 mg used in the treatment of leukemia as recommended in the label approved by FDA
Dec 28, 2018 : Shilpa Medicare Ltd has received U.S. Food and Drug Administration approval for its ANDA Irinotecan HCL Injection USP, 40 mg/2mL and 100 mg/5mL (20mg/mL) Single Dose Vials.Irinotecan injection is used in the treatment of patients with metastatic carcinoma of the colon or rectum.
Nov 13, 2018 :
• Shilpa Medicare Limited’s ANDA (#210291) for Dimethyl Fumarate Delayed Release Capsules 120 mg & 240 mg has been granted tentative approval by FDA on November 09, 2018.
• Dimethyl Fumarate Delayed Release Capsules 120 mg & 240 mg is a generic of TECFIDERA, used in the treatment of patients with relapsing forms of multiple sclerosis.
• This ANDA is a first to file submission made on NCE-1 date Mar 27, 2017. FDA review process was completed and got approval within a period of 19.5 months from the date of submission.
| Country | Approval Date | MAH | MA Status | MA Number |
|---|---|---|---|---|
| CZ | 10-Oct-18 | KHC, AT | Approved | 44/597/16-C, 44/598/16-C |
| Country | Approval Date | MAH | MA Status | MA Number |
|---|---|---|---|---|
| EE | 02-Oct-18 | KHC, AT | Approved | 973118, 973218 |
| Country | Approval Date | MAH | MA Status | MA Number |
|---|---|---|---|---|
| MT | 04-Sep-18 | KHC, AT | Approved | MA1280/00101, MA1280/00102 |
June 15, 2018: Shilpa Medicare Limited bagged the most prestigious Export Excellence Award on 15th June 2018 in recognition of the company's exemplary services in the export segment by Federation of Karnataka Chambers of Commerce and Industry (FKCCI).
Shilpa Medicare Limited bagged the most prestigious Export Excellence Award on 15th June 2018 in recognition of the company's exemplary services in the export segment by Federation of Karnataka Chambers of Commerce and Industry (FKCCI).
July 18 to July 26, 2016 :
• Formulation facility audited by USFDA from July 18 to July 26 and audit was successfully completed with Zero 483s.
• Both Raichur API facility (Unit-1 and unit-2) received GMP clearance from TGA - Australia
• Raichur API facility Unit-1 received GMP clearance from PMDA - Japan.
April 18, 2016: Our MD, Mr. Vishnukant Bhutada, is ranked # 35 as "India's Most Valuable CEO" in Mid-Size Companies Category (Sales between 250 Cr - 2,499 Cr) by Business World, a reputed business publication in India.
Feb 11, 2016Company both Unit-1 and 100% EOU Plant (Unit-2) API Manufacturing facility at Raichur, Karnataka has received compliance letter from USFDA
Nov 30, 2015 "Shilpa Medicare Ltd bagged UBM's "Excellence in R&D" Award (Company with turnover more than 500 Cr)."
"Shilpa Medicare Ltd bagged UBM's "Excellence in R&D" Award (Company with turnover more than 500 Cr)."
The Award was presented during the "India Pharma Awards 2015" Function at Mumbai on 30th November 2015. Read More
Oct 02, 2015 EUGMP received from Government of Upper Bavaria, Germany for Gemcitabine, Irinotecan HCL, Oxapplatin and Temozolomide to our Unit-1 at Raichur, Karnataka."
The plant was inspected by authority on 8th July 2015. Read More
Oct 09, 2015 EUGMP received from Government of Upper Bavaria, Germany for Bendamustine, Capecitabine and Melphalan to our Unit-2 at Raichur, Karnataka".
The plant was inspected by authority on 8th July 2015. Read More
Sep 23, 2015:  Shilpa Medicare Ltd bagged Pharmexcil's "Patent Award" for securing the most number of product patents. The Awards were presented during the "Awards Function for Exports and patents during 11th Annual General Meet of Pharmexcil at Hyderabad".
Shilpa Medicare Ltd bagged Pharmexcil's "Patent Award" for securing the most number of product patents. The Awards were presented during the "Awards Function for Exports and patents during 11th Annual General Meet of Pharmexcil at Hyderabad".
Sep 08, 2015:
Company 100% EOU Plant at Raichur, Karnataka has received GMP compliance from authority of Switzerland, Swissmedic for manufacturing of active substance Ambroxol Hydrochloride for three years.
The plant was inspected by authority between 18th March 2015 to 20th March 2015.
Aug 28, 2015:
Company SEZ Formulation unit at Jadcherla (Near Hyderabad), Telengana received GMP certification for both Human Medicinal Products & Human Investigational Medicinal Products from Republic of Slovenia.
The Jadcherla plant was inspected on 19-11-2014 by the Authorities.
Aug 28, 2015:
Company 100% EOU Plant at Raichur, Karnataka has received certification for CEP 2004-201 Ambroxol Hydrochloride.
The Raichur plant was inspected by European Directorate for the Quality of Medicines & Healthcare (EDQM) on 18-03-2015 to 20-03-2015.
Dec 19, 2014: Shilpa Medicare has benchmarked its quality processes against the world's best quality standards in various areas of operation. 1) ISO 9001:2008 for Quality Management System for SML unit-1 for period 13-12-2014 to 12-12-2017 and SML unit-2 100% EOU for period 09-12-2014 to 08-12-2017. 2) ISO 14001:2004 for Environment Management System for SML unit-1 for period 13-12-2014 to 12-12-2017 and SML unit-2 100% EOU for period 09-12-2014 to 08-12-2017. 3) Cofepris GMP Certificate, Mexico for Ambroxol, Nifedipine and Gemcitabine
Sep 25, 2013:  Shilpa Medicare observed Pharmacist's day in Raichur, Karnataka in collaboration with N.E.T Pharmacy College and V.L. College of Pharmacy.
Shilpa Medicare observed Pharmacist's day in Raichur, Karnataka in collaboration with N.E.T Pharmacy College and V.L. College of Pharmacy.
Jan 05, 2013: Shilpa Medicare bags first prize under pharma category of National Energy Conservation Award 2012
Shilpa Medicare bags first prize under pharma category of National Energy Conservation Award 2012
Jun 12, 2013: Shilpa Medicare commences functioning partially at its facility for manufacture of formulations, near Mahaboobnagar in Telangana, India.
Jun 27, 2013: Shilpa Medicare inks pact with MPP, Gilead for HIV/AIDS drugs
Sep 28, 2012:Hon'ble High Court of Andhra Pradesh has approved the scheme of amalgamation of Raichem Life Sciences Private Limited with Shilpa Medicare Ltd.
May 16, 2012:Tano Capital ups stake in Shilpa Medicare Ltd.
Jul 19, 2011: Shilpa Medicare has acquired controlling stake in Shilpa Therapeutics
